Dexamethasone
Dexamethasone Bottle [1]
One consequence of traditional antibiotic meningitis treatment is bacterial lysis which may lead to inflammation of the subarachnoid space, resulting in unfavorable neurological outcomes. Dexamethasone, an anti-inflammatory medication, has recently been introduced to combat these effects and can replace steroids in cases of adrenal insufficiency. Not only used in cases of meningitis, dexamethasone is commonly used to reduce edema in the spine and brain [2].
For more information on the basic information regarding dexamethasone, visit http://www.chemocare.com/bio/dexamethasone.asp
In the past decade, dexamethasone has been adopted as a popular adjuvant therapy for pneumococcal meningitis. It has since been implemented on a large scale in the Netherlands and is now commonly recommended for use in high-income countries [2]. However, studies vary on the degree and even the existence of its beneficial effects. One Cochrane review associated its use with lower mortality rates and lower rates of hearing loss, a disability which occurs in 22-30% of survivors of pneumoccal meningitis. On the other hand, "a recent meta-analysis of individual patient data of five randomized controlled trials" disagrees with these findings, claiming there to be "no effect of adjunctive dexamethasone in pneumococcal meningitis" [3]. Dr. Brouwer and his colleagues at the Center of Infection and Immunity Amsterdam (CINIMA) sought to review these findings in a 2006-2009 study of all Dutch patients over 16 years of age with pneumococcal meningitis. The outcomes of these patients were compared with those of a similar cohort from 1998-2002, before dexamethasone treatment was commonplace. Brouwer and his team discovered a decrease in fatality from 30% to 20% which they attributed to dexamethasone use after correcting for changes in disease severity and differences in case mix between the cohorts. However, they also found data indicating dexamethasone to be more effective in patients over 55 years of age, a finding which they cite as a possible cause of the conflicting results in randomized controlled trials which used patients of various ages. Further research will be required to support their suggestion that dexamethazone provides more benefit to middle aged and older individuals.
Dexamethasone use in infants is even more controversial. The American Academy of Pediatrics Committee on Infectious Diseases advises consideration of dexamethasone for infants at least six weeks of age with pneumococcal meningitis [4]. Nevertheless, Dr. Kim of the Johns Hopkins School of Medicine warns that dexamethasone may make it particularly difficult to monitor clinical responses to antimicrobial treatment including fever curves given that recurrence of fever occurs 28% more often when dexamethasone is administered. In addition, dexmethasone may inhibit vancomysin penetration into the CSF due to dexamethasone's anti-inflammatory properties, causing treatment failure. The drug has also been shown to cause neuronal injury in the form of hippocampal apoptosis in animals with pneumoccal and E coli meningitis. Dr. Kim recommends long-term cognitive and neuropsychological follow up studies on individuals receiving dexamethasone treatment.
For more information on the basic information regarding dexamethasone, visit http://www.chemocare.com/bio/dexamethasone.asp
In the past decade, dexamethasone has been adopted as a popular adjuvant therapy for pneumococcal meningitis. It has since been implemented on a large scale in the Netherlands and is now commonly recommended for use in high-income countries [2]. However, studies vary on the degree and even the existence of its beneficial effects. One Cochrane review associated its use with lower mortality rates and lower rates of hearing loss, a disability which occurs in 22-30% of survivors of pneumoccal meningitis. On the other hand, "a recent meta-analysis of individual patient data of five randomized controlled trials" disagrees with these findings, claiming there to be "no effect of adjunctive dexamethasone in pneumococcal meningitis" [3]. Dr. Brouwer and his colleagues at the Center of Infection and Immunity Amsterdam (CINIMA) sought to review these findings in a 2006-2009 study of all Dutch patients over 16 years of age with pneumococcal meningitis. The outcomes of these patients were compared with those of a similar cohort from 1998-2002, before dexamethasone treatment was commonplace. Brouwer and his team discovered a decrease in fatality from 30% to 20% which they attributed to dexamethasone use after correcting for changes in disease severity and differences in case mix between the cohorts. However, they also found data indicating dexamethasone to be more effective in patients over 55 years of age, a finding which they cite as a possible cause of the conflicting results in randomized controlled trials which used patients of various ages. Further research will be required to support their suggestion that dexamethazone provides more benefit to middle aged and older individuals.
Dexamethasone use in infants is even more controversial. The American Academy of Pediatrics Committee on Infectious Diseases advises consideration of dexamethasone for infants at least six weeks of age with pneumococcal meningitis [4]. Nevertheless, Dr. Kim of the Johns Hopkins School of Medicine warns that dexamethasone may make it particularly difficult to monitor clinical responses to antimicrobial treatment including fever curves given that recurrence of fever occurs 28% more often when dexamethasone is administered. In addition, dexmethasone may inhibit vancomysin penetration into the CSF due to dexamethasone's anti-inflammatory properties, causing treatment failure. The drug has also been shown to cause neuronal injury in the form of hippocampal apoptosis in animals with pneumoccal and E coli meningitis. Dr. Kim recommends long-term cognitive and neuropsychological follow up studies on individuals receiving dexamethasone treatment.
Glycerol
Glycerol, traditionally used as an osmotic diuretic to reduce raised tissue pressure, has also been shown to be an effective adjuvant therapy for meningitis [5]. Dr. Peltola and his team at the Helsinki University Central Hospital in Finland suggest that glycerol's ability to reduce neurolgoical sequelae and prevent deafness may be superior to that of dexamethasone. However, the combined effects of the drugs result in even better outcomes. Further benefits of the treatment, Peltola et al. note, include its oral administration and safety as well as its availability and low cost, making it an ideal adjunctive treatment in low income and resource-limited settings.
Hypothermia
Saline IV used for Hypothermia Treatment [6]
Hypothermia treatment has long been recognized for its neuroprotective effects, a fact which is a particularly important in meningitis which is closely associated with neuronal damage, encephalopathy, and ischemic stroke [7]. Mild hypothermia (32-34 ⁰C) has been suggested as an especially useful therapy for patients with severe pneumococcal meningitis, for whom complementary therapies such as corticosteriods have little effect. And it is these individuals with severe community-acquired bacterial meningitis, that Drs. Lepur, Kultesa, and Barsic decided to include in their 2010 study. They felt that where other complementary treatment methods failed in seeking only to reduce intracranial pressure, hypothermia would more directly target damage in cerebral blood flow (CBF) autoregulation, which they felt more greatly contributed to the unfortunate neurological conditions described above.
Hypothermia is induced via intravenous infusion of cold saline [7]. In decreasing body temperature, hypothermia causes a number of beneficial effects, including "reduced production of reactive oxygen and nitrogen species, inhibition of the neuroexcitatory cascade, a reduction in proinflammatory cytokine level, maintenance of blood-brain barrier integrity, decreased intracranial pressure, and finally decreased neuronal apoptosis" [7]. In the 2010 study mentioned above, patients were selected based on their low levels of cerebrovascular carbon dioxide reactivity (CO2R) measured by Transcranial Doppler, which Lepur, Kutlesa, and Barsic claim to be the best indicator of potential benefit from hypothermia treatment. This value continued to be measured throughout the course of the experiment. Application of hypothermia consistently resulted in a reduction in optic nerve sheath diameter (indicating decreased intracranial pressure) and improved CO2 reactivity. Norepinephrine was used to increase mean blood flow velocity and decrease lactate-oxygen index, indicating increased perfusion. It was determined that without hypothermia treatment, the disease would have been fatal in all 10 cases of illness. As it was, many patients died from complications existing prior to their treatment, including refractory intracranial hypertension and severe residual neurological deficits. However, two of the six survivors had complete neurological recovery. Large scale, randomized trials are recommended to determine the efficacy of hypothermia treatment in less severe cases of meningitis and whether damaged CBF autoregulation is an adequate metric for determining patient benefit from hypothermia treatment.
Hypothermia is induced via intravenous infusion of cold saline [7]. In decreasing body temperature, hypothermia causes a number of beneficial effects, including "reduced production of reactive oxygen and nitrogen species, inhibition of the neuroexcitatory cascade, a reduction in proinflammatory cytokine level, maintenance of blood-brain barrier integrity, decreased intracranial pressure, and finally decreased neuronal apoptosis" [7]. In the 2010 study mentioned above, patients were selected based on their low levels of cerebrovascular carbon dioxide reactivity (CO2R) measured by Transcranial Doppler, which Lepur, Kutlesa, and Barsic claim to be the best indicator of potential benefit from hypothermia treatment. This value continued to be measured throughout the course of the experiment. Application of hypothermia consistently resulted in a reduction in optic nerve sheath diameter (indicating decreased intracranial pressure) and improved CO2 reactivity. Norepinephrine was used to increase mean blood flow velocity and decrease lactate-oxygen index, indicating increased perfusion. It was determined that without hypothermia treatment, the disease would have been fatal in all 10 cases of illness. As it was, many patients died from complications existing prior to their treatment, including refractory intracranial hypertension and severe residual neurological deficits. However, two of the six survivors had complete neurological recovery. Large scale, randomized trials are recommended to determine the efficacy of hypothermia treatment in less severe cases of meningitis and whether damaged CBF autoregulation is an adequate metric for determining patient benefit from hypothermia treatment.
A New Meningococcal Vaccine for Africa
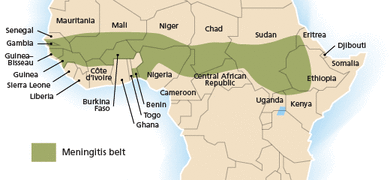
Map of Africa's Meningitis Belt [8]
There exists a stretch of 25 countries from Senegal to Ethiopia which make up Africa's Meningitis Belt [9]. In these and surrounding countries, over 450 million people are at risk for meningococcal meningitis. Fatality rates soar with little access to antibiotic treatment, as in 1996 and 1997 when a single epidemic caused 250,000 people to become ill and 25,000 to die. But other factors compound the impact of the disease. La Force and Okwo-Bele state in their Health Affairs article that "when medicine, nursing care, transportation to health services, lost wages, and other direct and indirect costs are tallied, a single case of meningitis may cost a Burkina Faso family about $90 - the equivalent of three to four months' income" [9]. The cost to the government is similarly high. In 2007, a meningitis outbreak cost Burkina Faso 5 percent of its health care budget, or $9.43 million. To cut healthcare costs and reduce the death toll, effective preventative care is an absolute necessity. Polysaccharide vaccines, which use the outer coatings of meningococcal bacteria to induce an immune response, are currently the standard method of prevention. But these vaccines are deficient in that they only provide two to three years of protection, do not block person-to-person transmission, and do not work effectively in infants and toddlers. The vaccine is also insufficiently produced, resulting in too little vaccine being distributed to countries in need too late.
Following the 1996-1997 epidemic, development of a new vaccine began which employed the concept of conjugating a protein to a polysaccharide antigen. Proteins used in prior research include diptheria and tetanus toxoids, toxins rendered non-toxic and used to stimulate an immune response via antibody production. The vaccines produced by these methods, LaForce and Okwo-Bele write, are both "more powerful and capable of providing longer-lasting protection" [9]. The Bill and Melinda Gates Foundation established the Meningitis Vaccine Project which partnered the WHO and the Program for Appropriate Technology in Health, or "PATH". The Meningitis Vaccine Project then went on to establish relationships with SynCo Bio Partners in Amsterdam to provide purified capsular sugar from Group A meningococci and the Serum Institute of India to produce the vaccine at a cost of $0.40, a price affordable for the affected African countries and low enough to allow for vaccination of birth cohorts, not simply as a reactive measure to stem epidemic spread. Clinical trials have confirmed the safety of the new conjugate vaccine and its ability to raise blood antibody levels to twenty times the levels brought on by the polysaccharide vaccine.
The vaccine was first employed in Burkina Faso, Niger, and Mali due to their high and sustained rates of meningitis [9]. On December 6, 2010 almost 3,000 vaccination teams were given 10 days to vaccinate eleven million people in Burkina Faso. Similar programs were initiated in Niger and Mali and by the end of 2010, 19.5 million people had been vaccinated in these three countries. The biggest hurdle for carrying out these massive vaccination programs is a financial one. While the GAVI Alliance (formerly 'the Global vaccine is Alliance for Vaccines and Immunization') is helping to provide financial aid for initial mass vaccination campaigns, countries interested in carrying out such campaigns are required by the Alliance to generate half of the required funds. In addition, GAVI aid ends after initial vaccination efforts, national and local governments will be responsible for generating continued funds to vaccinate newborns, once the WHO approves the vaccine for use in infants, expected in 2014.
Use of the conjugate vaccine within the "hyperendemic region" of meningitis belt, with a population of about 240 million, is expected to prevent 142,000 deaths and 284,000 disabilities over the next decade and save $350 million [9]. LaForce and Okwo-Bele caution that $475 million of additional funding will be required to realize the vaccine's promise "to eliminate epidemic meningitis from sub-Saharan Africa." Over the next few years, researchers will continue to gather data on the life-saving benefits of the vaccine in Burkina Faso, Niger, and Mali. The WHO will also continue to conduct trials on the safety of the vaccine for infants.
Following the 1996-1997 epidemic, development of a new vaccine began which employed the concept of conjugating a protein to a polysaccharide antigen. Proteins used in prior research include diptheria and tetanus toxoids, toxins rendered non-toxic and used to stimulate an immune response via antibody production. The vaccines produced by these methods, LaForce and Okwo-Bele write, are both "more powerful and capable of providing longer-lasting protection" [9]. The Bill and Melinda Gates Foundation established the Meningitis Vaccine Project which partnered the WHO and the Program for Appropriate Technology in Health, or "PATH". The Meningitis Vaccine Project then went on to establish relationships with SynCo Bio Partners in Amsterdam to provide purified capsular sugar from Group A meningococci and the Serum Institute of India to produce the vaccine at a cost of $0.40, a price affordable for the affected African countries and low enough to allow for vaccination of birth cohorts, not simply as a reactive measure to stem epidemic spread. Clinical trials have confirmed the safety of the new conjugate vaccine and its ability to raise blood antibody levels to twenty times the levels brought on by the polysaccharide vaccine.
The vaccine was first employed in Burkina Faso, Niger, and Mali due to their high and sustained rates of meningitis [9]. On December 6, 2010 almost 3,000 vaccination teams were given 10 days to vaccinate eleven million people in Burkina Faso. Similar programs were initiated in Niger and Mali and by the end of 2010, 19.5 million people had been vaccinated in these three countries. The biggest hurdle for carrying out these massive vaccination programs is a financial one. While the GAVI Alliance (formerly 'the Global vaccine is Alliance for Vaccines and Immunization') is helping to provide financial aid for initial mass vaccination campaigns, countries interested in carrying out such campaigns are required by the Alliance to generate half of the required funds. In addition, GAVI aid ends after initial vaccination efforts, national and local governments will be responsible for generating continued funds to vaccinate newborns, once the WHO approves the vaccine for use in infants, expected in 2014.
Use of the conjugate vaccine within the "hyperendemic region" of meningitis belt, with a population of about 240 million, is expected to prevent 142,000 deaths and 284,000 disabilities over the next decade and save $350 million [9]. LaForce and Okwo-Bele caution that $475 million of additional funding will be required to realize the vaccine's promise "to eliminate epidemic meningitis from sub-Saharan Africa." Over the next few years, researchers will continue to gather data on the life-saving benefits of the vaccine in Burkina Faso, Niger, and Mali. The WHO will also continue to conduct trials on the safety of the vaccine for infants.
SOURCES
[1] "Dexamethasone to prevent postoperative nausea and vomiting."Medicine & Science. N.p., 12 Aug. 2011. Web. 2 Dec. 2011. <http://medicine-science.com/dexamethasone-to-prevent-postoperative-nausea-and-vomiting/>.
[2] Gans, J. d., & de Beek, D. v. (2002). Dexamethasone in adults with bacterial meningitis. N Engl J Med,347, 1549-1556.
[3] Brouwer, M., Heckenberg, S., Gans, J. d., Spanjaard, L., Reitsma, J., & de Beek, D. v. (2010). Nationwide implementation of adjunctive dexamethasone therapy for pneumococcal meningitis. Neurology, 75(17), 1533-1539.-Colombini A, Bationo F, Zongo Z, Ouattra F, Jaillard P, Seini E, et al. Costs for households and community perception of meningitis epidemics in Burkina Faso. Clin Infect Dis. 2009; 49(10):1520-5.
[4] Kim, K. S. (2010). Acute bacterial meningitis in infants and children. The Lancet, 10(1), 32-42.
[5] Peltola, H., Roine, I., Bologna, R., Mino, G., Goyo, J., Lopez, E., et al. (2007). Adjuvant glycerol and/or dexamethasone to improve the outcomes of childhood bacterial meningitis: a prospective, randomized, double-blind, placebo-controlled trial. Clin Infect Dis, 45(10), 1277-86.
[6] Harris, Justin. "Training 1-24-08."elitefts. N.p., 28 Jan. 2008. Web. 2 Dec. 2011. <http://asp.elitefts.net/qa/training-logs.asp?qid=66800&tid=139 >.
[7] Lepur, D., Kutlesa, M., & Barsic, B. (2011). Induced hypothermia in adult community-acquired bacterial meningitis-more than just a possibility?. Journal of Infection, 62, 172-177.-Peltola, H., Roine, I., Bologna, R., Mino, G., Goyo, J., Lopez, E., et al. (2007). Adjuvant glycerol and/or dexamethasone to improve the outcomes of childhood bacterial meningitis: a prospective, randomized, double-blind, placebo-controlled trial. Clin Infect Dis, 45(10), 1277-86.Gans, J. d., & de Beek, D. v. (2002). Dexamethasone in adults with bacterial meningitis. N Engl J Med,347, 1549-1556.
[8] Control of epidemic meningococcal disease, WHO practical guidelines, World Health Organization, 1998, 2nd edition, WHO/EMC/BAC/98.3.
[9] LaForce, F. M., & Okwo-Bele, J. (2011). Eliminating epidemic group a meningococcal meningitis in africa through a new vaccine. Health Affairs, 30(6), 1049-1057.
[2] Gans, J. d., & de Beek, D. v. (2002). Dexamethasone in adults with bacterial meningitis. N Engl J Med,347, 1549-1556.
[3] Brouwer, M., Heckenberg, S., Gans, J. d., Spanjaard, L., Reitsma, J., & de Beek, D. v. (2010). Nationwide implementation of adjunctive dexamethasone therapy for pneumococcal meningitis. Neurology, 75(17), 1533-1539.-Colombini A, Bationo F, Zongo Z, Ouattra F, Jaillard P, Seini E, et al. Costs for households and community perception of meningitis epidemics in Burkina Faso. Clin Infect Dis. 2009; 49(10):1520-5.
[4] Kim, K. S. (2010). Acute bacterial meningitis in infants and children. The Lancet, 10(1), 32-42.
[5] Peltola, H., Roine, I., Bologna, R., Mino, G., Goyo, J., Lopez, E., et al. (2007). Adjuvant glycerol and/or dexamethasone to improve the outcomes of childhood bacterial meningitis: a prospective, randomized, double-blind, placebo-controlled trial. Clin Infect Dis, 45(10), 1277-86.
[6] Harris, Justin. "Training 1-24-08."elitefts. N.p., 28 Jan. 2008. Web. 2 Dec. 2011. <http://asp.elitefts.net/qa/training-logs.asp?qid=66800&tid=139 >.
[7] Lepur, D., Kutlesa, M., & Barsic, B. (2011). Induced hypothermia in adult community-acquired bacterial meningitis-more than just a possibility?. Journal of Infection, 62, 172-177.-Peltola, H., Roine, I., Bologna, R., Mino, G., Goyo, J., Lopez, E., et al. (2007). Adjuvant glycerol and/or dexamethasone to improve the outcomes of childhood bacterial meningitis: a prospective, randomized, double-blind, placebo-controlled trial. Clin Infect Dis, 45(10), 1277-86.Gans, J. d., & de Beek, D. v. (2002). Dexamethasone in adults with bacterial meningitis. N Engl J Med,347, 1549-1556.
[8] Control of epidemic meningococcal disease, WHO practical guidelines, World Health Organization, 1998, 2nd edition, WHO/EMC/BAC/98.3.
[9] LaForce, F. M., & Okwo-Bele, J. (2011). Eliminating epidemic group a meningococcal meningitis in africa through a new vaccine. Health Affairs, 30(6), 1049-1057.
